With intense regulatory pressure and zero tolerance for contamination or downtime, errors in pharma manufacturing environments have major consequences.
Despite this, decisions regarding renovations and upgrades in pharma facilities are still being made using outdated or incomplete data, hindering planning and putting projects at risk.
The complex and heavily regulated nature of pharma plants often leads to fragmented data and the lack of an up-to-date record of what currently exists.
A combination of dense, interconnected systems (HVAC, cleanrooms, piping etc); ageing infrastructure; and siloed data sitting with different teams in varying formats mean that planning renovations or upgrades in live production environments is particularly challenging for all involved.
Add to this the fact that change control is slow due to validation requirements and rework can stretch to 30% of construction budgets, threatening delivery timelines and investment costs, and it’s no wonder that pharma leaders are increasingly looking for ways to improve their data management so that decision making is based on real scenarios rather than assumptions.

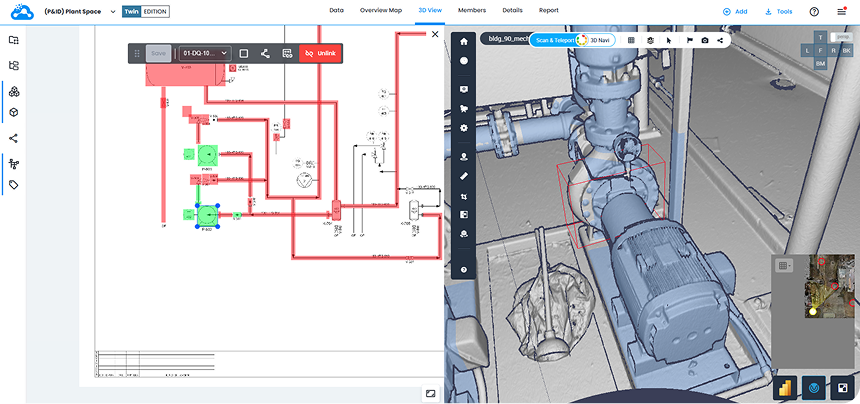
Digital twins have brought much needed clarity to pharma operations and Cintoo’s platform has become an important part of these workflows, providing teams with a single source of truth across asset lifecycles for improved understanding, collaboration and decision-making.
“Cintoo’s core features like 3D navigation and 3D scan-to-mesh provided strong value for our expert engineering users where precision is a must,” said a spokesperson from Arkema.
Embedding reality capture practices within the organization and using a platform like Cintoo to manage scan data, compare it with models and open up access to all stakeholders, vastly improves project workflows across the following areas:
Construction and fit-out: Capture as-built data in days, rather than weeks or months, and run clash detection before installation to identify issues ahead of time, reducing RFIs and change orders and avoiding cost overruns.
Operations and maintenance: Remote virtual walkthroughs and maintenance planning with precise spatial context minimize the need to access sensitive areas in-person and reduce downtime and risk.
Commissioning and qualification (CQV): Compare as-built data to the design to identify discrepancies and cut commissioning time by 30-50% by doing this virtually and reducing time on-site.
Safety, GMP and compliance: AI-powered detection of safety equipment, real-time asset tracking and audit-ready digital records bolster compliance and reduce regulatory risk.

By accurately capturing what exists, comparing this data to design models and feeding into digital twins, facilities management teams can transform operations and make significant time and cost savings.
During the design phase, digital modelling can lead to a third of undersized equipment being caught early. Scanning can be done in days instead of weeks, reducing time and resource and improving accuracy to millimetric levels.
Starting the build with better planning and foresight means rework can be slashed from 30% to less than 1%, saving millions of dollars per project. Accelerate commissioning by cutting down the required time by 30–50% and instill better coordination to eliminate 43% of on-site errors.
According to Critical Mass Consulting, "In the past three years, five major pharmaceutical facilities experienced start-up delays ranging from four to twelve months. The average cost impact? Over $20 million per facility from lost production revenue and remediation costs alone."
There’s also a myriad of ongoing operational benefits from fewer site visits reducing contamination risk to remote stakeholder collaboration and VR-based training enabling safer onboarding.

Reality-based digital twins should be viewed as an operational baseline for pharma companies, supporting continuous change and reconfiguration, enabling agile manufacturing models and creating a persistent, auditable record of assets. Thorough planning, full transparency and predictable project outcomes are essential in reducing risk and limiting downtime.
Are reality capture and digital twins already part of your GMP strategy? If not, experience your data in Cintoo today with a free trial - helping you leverage reality capture and digital twin insights to reduce RFIs, shore up compliance quickly, and align with GMP changes.